Varying Structures
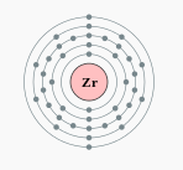
Zirconium is an element with five known isotopes. These isotopes are Zr-90, Zr-91, Zr-92, Zr-94 and Zr-96. Each known isotope has a relative abundance, or the percentage of an isotope occurring in a natural sample of an element. Relative abundance is also known as natural abundance. The relative abundance of Zr-90 is 51.45, Zr-91 has the relative abundance of 11.22, Zr-92 is 17.15, Zr-94 has 18.38 and Zr-96 has 2.8. An ion is an atom that has lost or gained valence electrons, and has a charge because of it. The ions it can make at +4 or -4. The plus four means that it can lose 4 valence electrons and the -4 means that it can gain four valence electrons. Electrons are negatively charged, which is why when you lose them they become positive, visa versa. The reason why zirconium can lose and gain valence electrons is because it has four, which is half of eight. Eight is the magic number for valence electrons, which means that all atoms want to either lose all of their electrons to help another atom have eight valence electrons in their outer shell, or take valence electrons from another atom to obtain eight valence electrons in its outer shell. A group is the number of columns across on the periodic table. The number of valence electrons of an element is always the same as the group number, so zirconium therefore has 4 valence electrons and is in group 4.The number of energy levels of zirconium is 5, and I know this because it is located in family five. Families are the rows across on the periodic table. The family number is always equal to the number of energy levels.
Zirconium is an element with five known isotopes. These isotopes are Zr-90, Zr-91, Zr-92, Zr-94 and Zr-96. Each known isotope has a relative abundance, or the percentage of an isotope occurring in a natural sample of an element. Relative abundance is also known as natural abundance. The relative abundance of Zr-90 is 51.45, Zr-91 has the relative abundance of 11.22, Zr-92 is 17.15, Zr-94 has 18.38 and Zr-96 has 2.8. An ion is an atom that has lost or gained valence electrons, and has a charge because of it. The ions it can make at +4 or -4. The plus four means that it can lose 4 valence electrons and the -4 means that it can gain four valence electrons. Electrons are negatively charged, which is why when you lose them they become positive, visa versa. The reason why zirconium can lose and gain valence electrons is because it has four, which is half of eight. Eight is the magic number for valence electrons, which means that all atoms want to either lose all of their electrons to help another atom have eight valence electrons in their outer shell, or take valence electrons from another atom to obtain eight valence electrons in its outer shell. A group is the number of columns across on the periodic table. The number of valence electrons of an element is always the same as the group number, so zirconium therefore has 4 valence electrons and is in group 4.The number of energy levels of zirconium is 5, and I know this because it is located in family five. Families are the rows across on the periodic table. The family number is always equal to the number of energy levels.
Atomic Composition
Zirconium is a transition metal, and has the atomic symbol of Zr. Its atomic number is 40, which means that it has 40 protons. The number of protons of an element is always equal to the atomic number. Protons are in the nucleus of the atom, and they are in there with the neutrons. Protons are positively charged, and neutrons do not have a charge. The purpose of neutrons is to hold the protons together. The number of neutrons that zirconium has is 51. Also, zirconium has the amu, or atomic mass unit, of 91.224.